
by Vincent Franceschi
*A micrometer is 1/1,000,000, or 1 x 10-6 meter. 1. A water lettuce plant (Pistia stratiotes) taken with a macro lens and 35 mm camera. This is the level of resolution we commonly use to observe the world around us with our eyes. We can clearly see that the plant is made of various parts (organs), and we can deduce certain biochemical features based on our general knowledge of plants. (For example, it is green and therefore likely to exhibit photosynthesis.) But we can't see what the plant really looks like or how it functions at the tissue or cellular level without the aid of additional tools. For example, how does this plant cope with the relatively large amounts of calcium it is exposed to in its environment? Calcium is an important nutrient that is tightly regulated at the cellular and subcellular level in all living organisms. 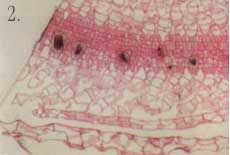 2. Here is a one-micrometer-thick section through a leaf of a water lettuce plant. The plant has been exposed to radioactive calcium, then processed to allow for visualization of where calcium has accumulated at the tissue and cellular level. The radioactive calcium appears as small dark particles. Calcium is accumulating in specific cells, which appear grayish or black among the non-accumulating cells stained red/pink. Using light microscopy with this technique, we can see that the plant has specialized cells designed for regulating calcium levels in the leaf. This view does not tell us much about the special features of this cell type, though. 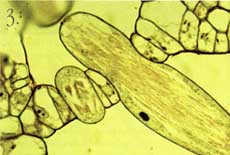 3. This section through the leaf has been photographed at higher magnification with a light microscope and gives some detail on the specialized calcium-accumulating cells, which we call idioblasts. The idioblast is elongated relative to surrounding cells and has calcified material in the vacuole. 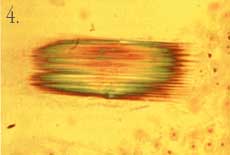 4. Calcified material often is crystalline in nature and can be viewed using polarizing filters with the light microscope. This photograph shows the material in the vacuole is clearly crystalline in nature and may occur as a bundle of individual crystals. However, because of the relatively short depth of field of the light microscope objective lens, it is difficult to get a clear view of the individual crystals.  5. By using a scanning electron microscope (SEM), a three-dimensional image of the crystal can be obtained. The SEM has much greater depth of field and also high resolution through the use of electrons (very short wavelength compared to visible light) for image formation. In this picture we see an idioblast that has been cleaved open, revealing the crystals in the vacuole. The crystals are clearly seen as four-sided structures with pointed ends. The SEM view also demonstrates that they are packed into a bundle within the idioblast. 6-8. While calcification is common in animals (bones, teeth, etc.), not much is known about such processes in plants. The crystals in water lettuce are known to be made of calcium oxalate, and it is of interest to determine how they grow within the idioblasts. Using plants exposed to radioactive calcium, followed by specialized preparation techniques, we can show that the cell carefully controls crystal growth. We also find that growth of individual crystals is bi-directional. A laser scanning confocal microscope was used to generate the following images, which reconstruct crystal growth in water lettuce crystal idioblasts. The silver grains produced by decay of the radioactive calcium are imaged by laser light scattered from them, and the tissue section is imaged by the laser light transmitted through it. The confocal microscope can then combine the two images to give a "map" of radioactive calcium at the subcellular level. Thus, microscopes can be used to show dynamic events at the cellular level.  6. A very young developing idioblast with very small crystals.  7. A slightly older idioblast showing that the middle of the crystals is no longer adding calcium (not growing) while growth is clearly occurring from both ends of the crystals within the bundle.  8. A nearly mature idioblast showing growth occurring only at the very tips of the long crystals within the vacuole.  9. It would be interesting to see what structures within the idioblasts help regulate calcium accumulation. However, the resolution of the light microscope is limited by the wavelength that is possible to use for illumination, and very small structures are not clearly resolved. By using electrons that have very short wavelengths, the transmission electron microscope (TEM) can give tremendous improvement in resolution. This picture of an idioblast was taken with a TEM at relatively low magnification. However, a trained cell biologist will see that the idioblast cell has some very different features from a "typical" cell, including an abundance of endoplasmic reticulum (ER), the dark lines running through the cytoplasm. This organelle is known to be involved in calcium transport. In order to get a TEM image, very thin sections have to be made (60 nanometers*), while this cell is about 100 micrometers long. Thus, it is sometimes difficult to tell if the image is representative of the entire cell without much additional work.  10. Cellular probes developed for fluorescence light microscopy can be used to overcome the problem of determining if an image seen in TEM is representative of the entire cell. Although the resolution will not be as good as with TEM, in combination with laser scanning confocal microscopy entire cells can be imaged at fairly high resolution and in a living state! This image shows the distribution of endoplasmic reticulum in cells within a living piece of water lettuce leaf as seen with the confocal microscope. The intensity of fluorescence is a direct indication of the relative abundance of ER in the cells. The very bright green cells are all young crystal idioblasts. This technique clearly demonstrates that this cell type has large amounts of ER compared to a typical leaf cell, thus confirming the impression given by the TEM images.  11. This confocal microscope image of a mature crystal idioblast shows that ER occurs in large patches throughout the cell, which is different from the rather uniform distribution of this organelle seen in very young cells. This has some implications concerning physiological activity of the mature idioblasts. *A nanometer is 1/1,000,000,000, or 1 x 10-9 meter. Vince Franceschi is professor of botany and director of the Electron Microscopy Center. He studies plant structure-function relationships at the tissue, cellular, and subcellular levels. Photographs by Vince Franceschi and his graduate studentsTodd Kostman and Valerie Lynch-Holm. |